Paracervical block needle
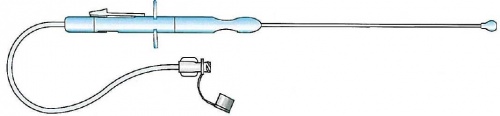
Paracervical block needle
Device designed and realized for an easy, safe, reliable and successflull execution of the uterine paracervical block. It consists of:
-
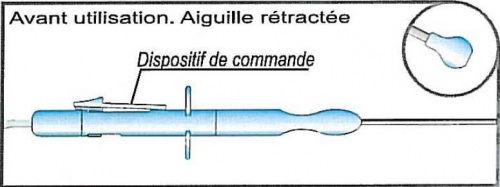
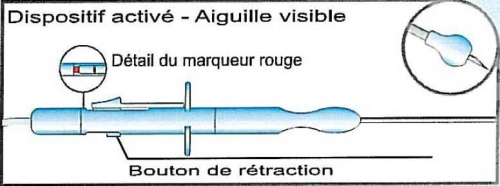
A handle at the proximal end of which a slide mechanism is placed to permit a safe and precise release of the needle tip outside a protective sheath; a red marker indicates the right and definitive release of the needle tip (this marker is hidden by the slide mechanism when at rest). The needle tip will return inside the protective sheath pushing the button placed just at the opposite side of the slide mechanism.
-
A stainless steel cannula (protective sheath) inside which the needle to perform the procedure is placed. The distal end of this cannula presents a protective plastic ball, so it is possible to ensure a steady and atraumatic contact with the junction of the cervix and the vagina
-
20 Ga, oblique tip stainless steel needle. The release of the needle tip outside the protective plastic ball is 4 mm (and the red marker appears)
-
PVC, extension line, 200 mm. length with female Luer lock and cap.
- Caracteristics
- Detail
- Related products
| Order number | Needle size | Total Length (extension excluded) | Extension length | Package q.ty |
| 2-PB02N | 20Ga (0,89mm) | 270mm | 200mm | 10 |
-
Sterilized by ethylene oxide (EtO).
-
ISO11135-1 validated process.
-
Shelf life : 36 month (3 years). The device is single use not resterilizable.
-
EtO residual : In conformity to harmonized international standard ISO10993-7
-
Biocompatibily : All the materials used for manufacturing are in compliance with ISO 10993-1 / USP Class VI requirements. The device is LATEX FREE.
No related product
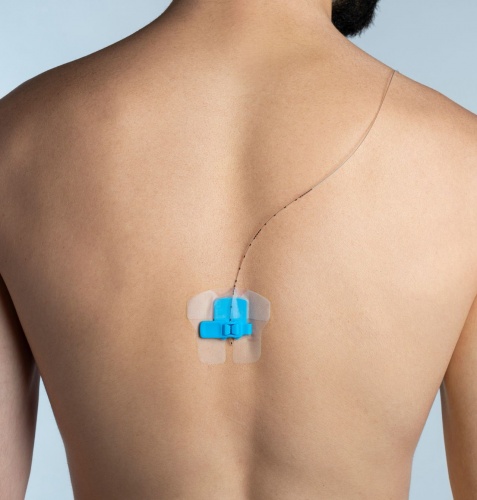
FlexGrip EPI
Patch for the stabilization for EPI/PNB
Sterile, single use patch for the catheter stabilization
Designed for the stabilization of Plexus & epidural catheters, spinal neuro modulation electrodes (pain management),
harpoons (breast cancer), CVC & midlines.
A ‘1 size fits all’ version which accepts any catheter between 16 - 20 Gauge (maximum 5Fr).
Easy to use,
very firm fixation,
repositionable,
low profile and soft material for comfort and high performing adhesive for safe and secure skin fixation
- Caracteristics
- Detail
- Related products
Ref : EP02
Composition
Hypoallergenic tapes - medical grade acrylic adhesive
Medical grade TPE, blue color
Sterilization by gamma radiation
Composition
Hypoallergenic tapes - medical grade acrylic adhesive
Medical grade TPE, blue color
Sterilization by gamma radiation
No related product